Asthma is a chronic disease with prominent heterogeneity in the pattern of underlying disease mechanisms, disease severity, clinical features, and responsiveness to specific treatments. Diagnosis of asthma is predominantly made with clinical history and examination. While the majority of asthmatic patients are controlled by standard pharmacological protocols of bronchodilators, a significant subgroup has very limited therapeutic options, representing a major unmet need. This limitation can be a probable result of underdiagnosis, misdiagnosis, or overdiagnosis of asthma.1

1) The usual diagnosis of asthma involves a very detailed history of similar
issues in the family, lifestyle, medications, and exposure to allergens like
pet hair, pollen, dust mites etc. The information of usual exacerbation timings
and climatic conditions is also of importance.
2) If the symptoms are atypical to asthma, the physician most likely recommends
X-ray imaging of the chest, sinus X-ray imaging, sputum analysis, and allergy
tests to rule out similar pathologies.
1) Lung function testing by spirometry2:
● Most easily accessible test for diagnosis of asthma in a clinical setting.
● The presence of an obstructive picture, with a ratio of FEV1/FVC <70%, and
associated reversibility following administration of bronchodilator is a strong
indicator of asthma as a diagnosis.
● If an obstructive spirometry result is present; it is recommended that all
adults aged 17 and over, should undergo a bronchodilator reversibility test. An
improvement of ≥12% in FEV1 and an increase in ≥200 ml may be considered a
positive test of reversible airflow obstruction.
● However, one of the key issues about the use of spirometry to help diagnose
asthma is the absolute requirement for its correct usage and interpretation.3
● Interpretation of spirometry results varies even between specialist lung
function laboratories, with a lack of standardisation in relation to
definitions for the lower limit of normal.4
● Although confirmation of reversible airflow obstruction is as important for a
diagnosis of asthma in children as in adults, the practical application of
spirometry in children is even more of a challenge.
● In children below 17 years of age, the test being comfortable and co-operated
for by children is rare.5
● Another critical issue for children is that when measured during stable
disease, spirometry is frequently normal, even in those with severe disease.6
2) Use of exhaled nitric oxide:
● As the fundamental pathophysiology underlying asthma incorporates
eosinophilic airway inflammation coupled with reversible airflow obstruction,
exhaled nitric oxide is considered additive to measures of lung function as an
indirect marker of airway inflammation.8
● If a value of 40 parts per billion (ppb) or higher is measured in a
patient with suspected asthma, this is considered a positive result, and
strongly supports asthma.9
3) Sputum eosinophil:
● In adult patients, the main use of sputum eosinophils has been to guide
management and tailoring of therapy to achieve a reduction in exacerbations,
rather than to make the diagnosis of asthma.10
4) Blood eosinophil:
● While interpreting blood eosinophils for asthma diagnosis, the very high
counts (>500 cells/mcl) have a high certainty of an associated airway
eosinophilia (not necessarily asthma), but for values <410 cells/mcl, the
relationship between blood and airway eosinophils becomes less clear. In such
cases, it is important to consider the overall clinical picture and all
possible factors that might affect blood eosinophil counts.11

NICE guidelines (2017)12
a) For children under 5 with suspected asthma, treat symptoms based on
observation and clinical judgement, and review the child regularly. If they
still have symptoms when they reach 5 years, carry out objective tests.
If a child is unable to perform objective tests when they are aged 5:
● Continue to treat based on observation and clinical judgment
● Try doing the tests again every 6 to 12 months until satisfactory results are
obtained
● Consider referral for specialist assessment if the child repeatedly cannot
perform objective tests and is not responding to treatment.
b) Offer a FeNO test to adults (aged 17 and over) if a diagnosis of asthma is
being considered.
Consider a FeNO level of 40 parts per billion (ppb) or more as a positive test.
However, consider the effects of smoking on false-positive values.
c) Offer spirometry to adults, young people, and children aged 5 and over if a
diagnosis of asthma is being considered. Consider a forced expiratory volume in
1 second/forced vital capacity (FEV1/FVC) ratio of less than 70% (or below the
lower limit of normal if this value is available) as a positive test for
obstructive airway disease (obstructive spirometry).
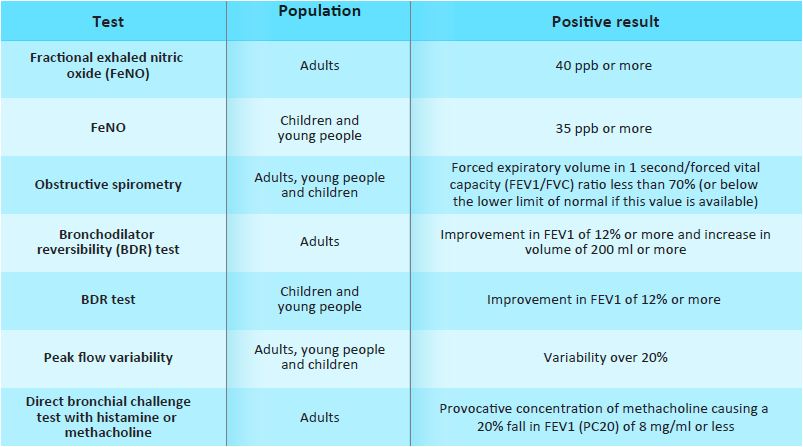
A multifactorial approach is
of utmost importance while diagnosing asthma, in order to avoid any
misdiagnosis, underdiagnosis, and overdiagnosis. Consideration of lung function
testing with spirometry along with FENO (fractional exhaled nitric oxide) can
be helpful. Better diagnosis will help in planning the management efficiently.
References: